A major goal of the O’Brien Kidney Center is to translate basic science research into effective diagnostic and therapeutic strategies that will improve the lives of patients by interrupting the pathogenesis of chronic kidney disease and its attendant high risk of cardiovascular disability and death.
Over the past 30 years, a large investment in biomedical research has led to substantial improvements in patient care and extended life in the general population. The Clinical and Translational Research Core is designed to encourage new collaborative efforts that tear down traditional silos between basic and clinical investigators. The primary objective is to support the highest quality clinical and translational research in kidney disease and associated cardiovascular disease by funded Center Investigators.
In addition to study populations available to investigators through their own initiatives, an important aspect of the O’Brien Kidney Center is access to the Dallas Heart Study population, a unique resource available at U.T. Southwestern Medical Center.
Distinctive Features of the Clinical and Translational Research Core
Traditionally, human renal and cardiovascular physiology studies have been performed on small selected study samples, while human genetic epidemiology studies have been conducted in large populations without physiological measurements, i.e., without intermediate phenotypes. Our Core is designed to combine state-of-the-art comprehensive human genetics with detailed state-of-the-art phenotyping of subjects drawn from unique study samples with unprecedented minority representation (below).
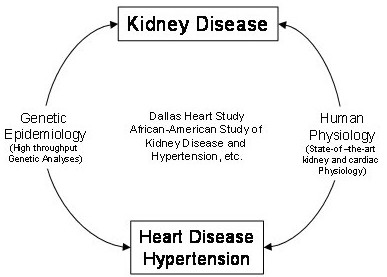
In this regard, a major strength of the Clinical and Translational Research Core is the availability of the Dallas Heart study—a large multiethnic population-based cohort with over 50 percent of the participants being African American—and the longitudinal cohort of the African American Study of Kidney Disease and Hypertension (AASK).
The Clinical and Translational Research Core will coordinate the recruitment of study participants for O’Brien Kidney Center.
Center Investigators and provide them with state-of-the-art phenotypic measurements of human renal and cardiovascular structure and function. Unique capabilities range from microelectrode recordings of sympathetic action potentials to proton magnetic resonance spectroscopic measurements of renal and cardiac steatosis – specific pathophysiological processes that could provide a common mechanistic explanation for the remarkably tight link between cardiac and renal disease as well as identifying novel therapeutic drug targets for early intervention sufficiently early to prevent (rather than merely delay) clinical endpoints such as end-stage renal disease, stroke, myocardial infarction and death.
Services
In addition to study populations available to investigators through their own initiatives, an important aspect of the O’Brien Kidney Center is access to the Dallas Heart Study population, a unique resource available at U.T. Southwestern Medical Center.
| Name | Role | Phone | Location | |
|---|---|---|---|---|
| Orson W. Moe, M.D. |
O'Brien Kidney Center Director
|
orson.moe@utsouthwestern.edu
|
||
| Wanpen Vongpatanasin, M.D. |
Clinical and Translational Research Core Co-Director
|
214-648-2103
|
wanpen.vongpatanasin@UTSouthwestern.edu
|
|
| Robert Toto, M.D. |
Clinical and Translational Research Core Co-Director
|
214-648-2600
|
robert.toto@UTSouthwestern.edu
|
| Price List |
| ► Patient Management (1) | |||
| Name | Description | Price | |
|---|---|---|---|
| Study participant recruitment and management |
|
Inquire | |